COVID-19 VACCINE INFORMATION FOR THE IMMUNOCOMPROMISED

Covid-19 Vaccine:
Booster Dose
For individuals who are NOT immunocompromised.
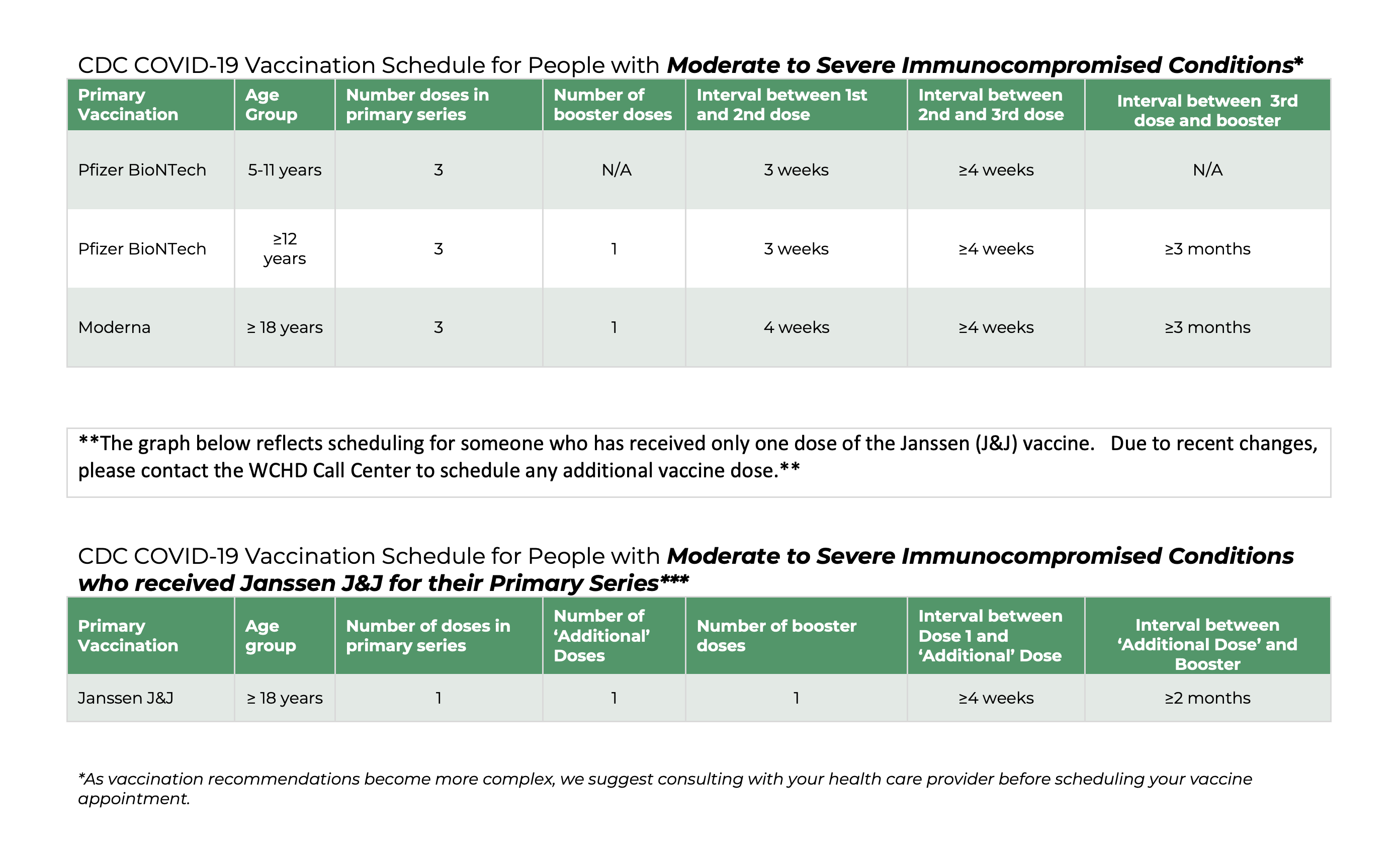
COVID-19 VACCINE:
THIRD DOSE & BOOSTER
for IMMUNOCOMPROMISED
COVID-19 VACCINE:
BOOSTER DOSES
For individuals who are NOT immunocompromised.
The Will County Health Department, Illinois Department of Public Health (IDPH), and Centers for Disease Control (CDC) is recommending anyone 12 years and older get a COVID-19 vaccine booster five months after completing their primary, two-dose series of Moderna or Pfizer, or two-months after receiving the one-dose Johnson & Johnson vaccine.
Immunocompromised? CLICK HERE.
Moderna
Booster Dose
WHO IS ELIGIBLE:
Individuals 18 years and older
DOSAGE:
½ dose
WHEN ARE YOU ELIGIBLE:
At least five-months after the initial two-dose series was administered
WANT TO SWITCH BRANDS?
Yes, you can receive a single booster dose of Moderna as long as you completed a one or two-dose series with a different COVID-19 vaccine.
Pfizer
Booster Dose
WHO IS ELIGIBLE:
Individuals 12 years and older
DOSAGE:
Full Dose
WHEN ARE YOU ELIGIBLE:
At least five-months after the initial two-dose series was administered
WANT TO SWITCH BRANDS?
Yes, you can receive a single booster dose of Pfizer as long as you completed a one or two-dose series with a different COVID-19 vaccine.
Johnson & Johnson
Booster Dose
WHO IS ELIGIBLE:
Individuals 18 years and older
DOSAGE:
Full Dose
WHEN ARE YOU ELIGIBLE:
At least two-months after a single dose primary vaccination was administered
WANT TO SWITCH BRANDS?
Yes, you can receive a single booster dose of Johnson & Johnson as long as you completed a two-dose series with a different COVID-19 vaccine.